AMS Fall Science Club 2016
Week 1: Slime
In our first week we learned about polymers, which are special molecules made of long, repeating units, sort of like beads on a necklace. These are very useful molecules in our modern world. Materials made from polymers tend to be very tough, stretchy, and sometimes glassy. Some natural polymer examples are rubber, wool, silk, proteins and DNA. Some synthetic (man-made) polymers are various kinds of plastic, nylon and synthetic rubber.
In Science Club we used a common polymer called white glue, and mixed it with weak boric acid (Borax powder dissolved in water). The boric acid acts as a "cross linker" to hook together the long polymer molecules, producing a stretchy substance called SLIME! Check out the videos below:
In Science Club we used a common polymer called white glue, and mixed it with weak boric acid (Borax powder dissolved in water). The boric acid acts as a "cross linker" to hook together the long polymer molecules, producing a stretchy substance called SLIME! Check out the videos below:
| Directions: How to Make Slime and Flubber |
Week 2: Flubber
|
Flubber is another polymer, similar to slime. It also uses boric acid as a cross linker, but instead of white glue, we use polyvinyl alcohol. This is a little harder to find- you won't see it in the grocery store- but you can find it on Amazon or at TeacherSource.com, among other places.
The slideshow above gives directions for making flubber as well as slime. Due to flubber's translucent nature, there are more options for creativity, including glitter or glow-in-the-dark (you can also buy glow powder online). Also at right you can find the answer keys for the Science and Math brainteaser sheets handed out in Club. |
| ||||
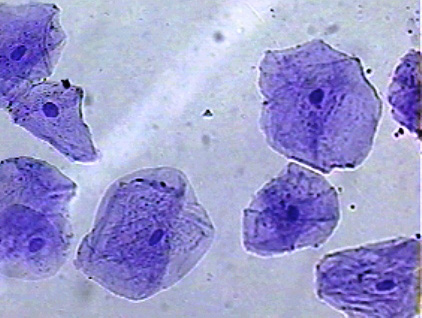
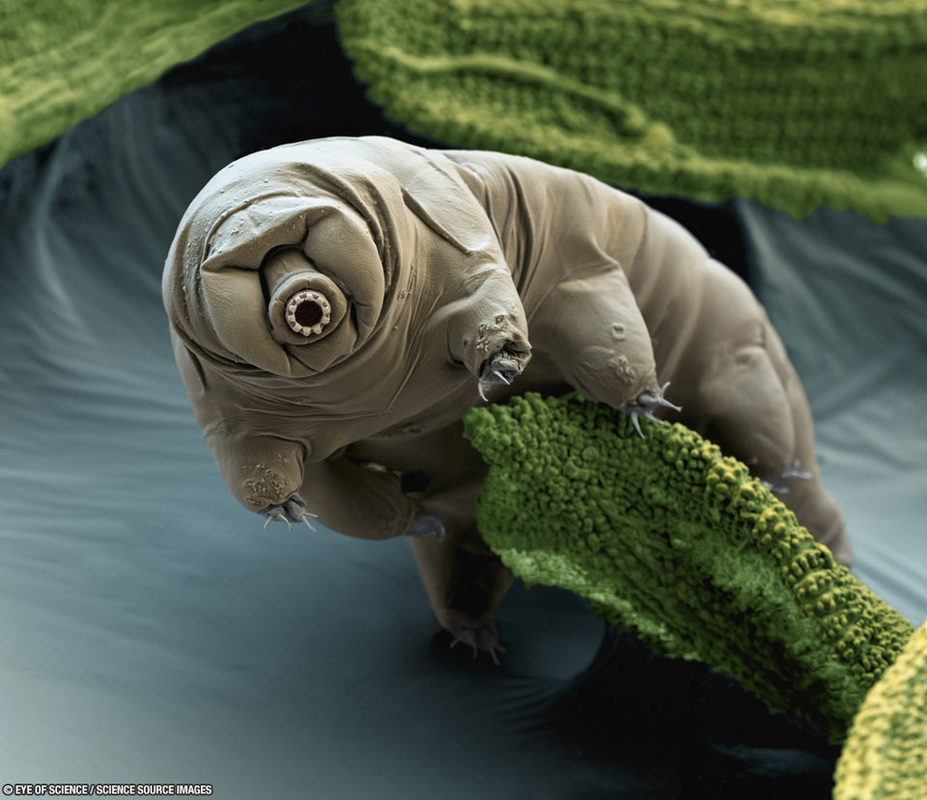
Weeks 3 and 4: Microscopes and Water Bears
|
We spent these two weeks learning how to use microscopes, and observing various items, both living and non-living. Students looked at onion cells, then collected their own "cheek" cells. We also learned about some microorganisms, especially the cute and cuddly water bears (tardigrades). Here is a link to a youtube video we watched which explains a little more about these fascinating and ancient animals.
Below, you can find an article I wrote with more information about tardigrades, and also a video of Science Club students exploring microscopes. |
Weeks 5 and 6: Building Telescopes, Exploring Astronomy

These telescopes should be perfect for viewing the moon, especially when it is only partially lit by the Sun. If you aim right at the "terminator"- the dividing line between light and dark on the moon, you should be able to see some mountains and craters on the moon.
In week 6, students watched a slideshow about "What You Can See With Your Telescope" (attached). Then we watched a really great iMax video, "Cosmic Voyage", which does an excellent job of conveying the scale and majesty of the cosmos. This amazing film is available to watch free online at this link.
If you are interested in knowing what is "out there" in the night sky at different times of the year, here are a few links that are updated daily with the latest recommended objects for telescope viewing.
EarthSky.org
HeavensAbove
Sky and Telescope
There are also several fantastic astronomy apps for smartphones or tablets, which can easily identify whatever you can see in the night sky, just by pointing your device's camera at the sky. Some good ones I have used are SkyView, Star Walk, Starmap, and pUniverse (Pocket Universe).
|
| ||||
Week 7 and 8: "Ten Things to Know About Air"
We investigated "t0 Things to Know About Air", including:
1. Air exists
2. Air is hard to see, feel and smell (but not impossible!)
3. Air takes up space
4. Air has weight (mass): 14.7 pounds per square inch, at Earth's surface
5. Air is compressible
6. Air is made of particles called molecules
7. The molecules can move closer together (contract) or farther apart (expand)
8. There are different kinds of air molecules, like oxygen, nitrogen or carbon dioxide
9. Air is a gas, but can change to other phases (solid, liquid or plasma)
10. A fluid that's moving has less pressure
Each of these ideas were demonstrated, with student help, as seen in the video below.
1. Air exists
2. Air is hard to see, feel and smell (but not impossible!)
3. Air takes up space
4. Air has weight (mass): 14.7 pounds per square inch, at Earth's surface
5. Air is compressible
6. Air is made of particles called molecules
7. The molecules can move closer together (contract) or farther apart (expand)
8. There are different kinds of air molecules, like oxygen, nitrogen or carbon dioxide
9. Air is a gas, but can change to other phases (solid, liquid or plasma)
10. A fluid that's moving has less pressure
Each of these ideas were demonstrated, with student help, as seen in the video below.
Week 9: Cartesian Divers
Students built Cartesian divers, which are made from a pipette (eye dropper) which contains just enough water to barely float in a large, full bottle of water. By squeezing the bottle of water, we can force a bit of extra water into the pipette, increasing its density and causing it to sink. After adding a few creative touches, students went home with some colorful, fun science toys!
By the way, Cartesian divers are named for the great mathematician and philosopher, Rene Descartes- yes, the same guy who taught us how to graph points on a "cartesian plane" using x and y coordinates. According to legend, Descartes was the first inventor of a Cartesian diver toy way back in the 1600's- although I very much doubt he used 2-liter pop bottles!
By the way, Cartesian divers are named for the great mathematician and philosopher, Rene Descartes- yes, the same guy who taught us how to graph points on a "cartesian plane" using x and y coordinates. According to legend, Descartes was the first inventor of a Cartesian diver toy way back in the 1600's- although I very much doubt he used 2-liter pop bottles!
Week 10: Immiscible Liquids

We explored the properties of "immiscible" liquids (liquids that don't easily mix), and also investigated the effect of adding a "surfactant" chemical (soap), which causes the liquids to become much more friendly with each other.
The students made two projects. In the first, they added to a 1-liter bottle two very immiscible liquids: mineral oil (aka baby oil) and water. If the bottle is filled to the brim and sealed, this produces a mesmerizing toy called a Bubble Bottle or Wave Bottle.
For the second project, we carefully poured several liquids into a new 1-liter bottle. Some of these liquids are quite immiscible (such as water and corn oil), while others are miscible (capable of mixing) if the bottle is shaken, such as corn syrup or rubbing alcohol.
Because of the varying densities of these liquids, they can "stack up" according to weight in the container.
The students made two projects. In the first, they added to a 1-liter bottle two very immiscible liquids: mineral oil (aka baby oil) and water. If the bottle is filled to the brim and sealed, this produces a mesmerizing toy called a Bubble Bottle or Wave Bottle.
For the second project, we carefully poured several liquids into a new 1-liter bottle. Some of these liquids are quite immiscible (such as water and corn oil), while others are miscible (capable of mixing) if the bottle is shaken, such as corn syrup or rubbing alcohol.
Because of the varying densities of these liquids, they can "stack up" according to weight in the container.
Week 11: Visible and Invisible Light
We learned about the different kinds of light in the electromagnetic spectrum: the visible light (red, orange, yellow, green, blue, indigo and violet) and the invisible-but VERY USEFUL- kinds of light (ultraviolet, infrared, radio waves, microwaves, x-rays, gamma rays). With the help of an ultraviolet light source (a "black light"), we had a hand-washing contest, and students found out just how difficult it is to wash ALL parts of your hands. Finally, we learned how the human eye can be "tricked" into mixing different colors of visible light that are moving rapidly. To demonstrate this, students built spinning color wheels, as seen in the clips below.
Week 12: Optical Illusions
We looked at different ways that different optical illusions can "trick" our eyes and brains. Here is a link to some of the online optical illusions we tried.
Students constructed the dragon/T-rex 3-d optical illusion that many have seen on the internet (here's a Youtube video example).
Students then made their own optical-illusion spinning wheels. This is done by hot-gluing a marble into the hole of an old CD disk. When an optical illusion pattern is placed over the CD, you can spin it with the marble touching a flat surface, and it will go for a very long time. Here is a video explanation from the source where I found this idea, a gentleman named Arvind Gupta, who creates all kinds of toys from recycled materials. (Check his website, www.arvindguptatoys.com for lots of other awesome ideas!)
The video below shows some examples of the illusions we made in class.
Students constructed the dragon/T-rex 3-d optical illusion that many have seen on the internet (here's a Youtube video example).
Students then made their own optical-illusion spinning wheels. This is done by hot-gluing a marble into the hole of an old CD disk. When an optical illusion pattern is placed over the CD, you can spin it with the marble touching a flat surface, and it will go for a very long time. Here is a video explanation from the source where I found this idea, a gentleman named Arvind Gupta, who creates all kinds of toys from recycled materials. (Check his website, www.arvindguptatoys.com for lots of other awesome ideas!)
The video below shows some examples of the illusions we made in class.
Week 13: Exploring Liquid Air
Liquid Nitrogen is simply air that has had so much heat removed that the gas condenses to a liquid form of matter. At this point, the oxygen and nitrogen liquids can be separated into different containers and stored in super-insulated containers. My giant thermos (called a "dewar") holds 20 liters of liquid nitrogen. When you see the liquid pouring out, it is right at its boiling point, which is -320° F.
Liquid nitrogen is mostly used as a way to make things extremely cold. This is useful for such things as quickly freezing food, or for freezing metal to make it shrink. For instance, you can make a metal screw that is slightly too large for its hole; freeze the screw to shrink it so it fits in the hole; then when the screw "warms up" it will expand and lock into the hole permanently.
We used liquid nitrogen to turn the air inside balloons to liquid; to freeze various vegetables and flowers (and a hot dog "finger"); and especially to make some really awesome ice cream. The video below shows some of the "cryogenic" fun in our last meeting of the Fall Science Club.
Liquid nitrogen is mostly used as a way to make things extremely cold. This is useful for such things as quickly freezing food, or for freezing metal to make it shrink. For instance, you can make a metal screw that is slightly too large for its hole; freeze the screw to shrink it so it fits in the hole; then when the screw "warms up" it will expand and lock into the hole permanently.
We used liquid nitrogen to turn the air inside balloons to liquid; to freeze various vegetables and flowers (and a hot dog "finger"); and especially to make some really awesome ice cream. The video below shows some of the "cryogenic" fun in our last meeting of the Fall Science Club.